Statins represent a class of drugs extremely useful for reducing blood cholesterol values in people predisposed to familial hypercholesterolemia, an important risk factor for atherosclerosis, a very dangerous pathological condition for heart and arterial health.
Simvastatin introduction
Hypercholesterolemia affects millions of people worldwide, and is the main risk factor for cardiovascular disease, especially in industrialized countries.
High levels of LDL (bad cholesterol) and fat (in the form of triglycerides) in the blood are associated with a higher incidence of cardiovascular disease, such as heart attack and stroke. Scientific studies show that oxidized deposits of cholesterol and transport proteins (LDL) along the walls of the arteries are the main cause of atherosclerosis, a chronic inflammatory disease that can cause obstruction of blood vessels, or detachment of blood clots.
Low-fat diets, such as the Mediterranean diet, and regular aerobic sporting activity are associated with a reduction in blood fat, and a reduction in death from cardiovascular accidents.
However, there are particular physiological conditions, such as diabetes or familial hypercholesterolemia, in which dietary remedies, a low-fat diet and physical activity are not sufficient to reduce plasma cholesterol and triglycerides. In these cases it is necessary to resort to the use of lipid-lowering drugs. Among these the statins they are the most common and prescribed.
Statins also have further beneficial effects on the cardiovascular level such as antioxidant, anti-inflammatory and antithrombotic effects (Di Garbo and colleagues 2000, Albert and colleagues 2001, Liao and Laufus 2005).
There simvastatin (Fig. 1), also known by the trade names of Zocor®, Sivastin®, Sinvacor®, is the precursor to all statins.
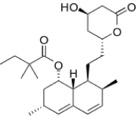
Fig. 1 - Molecular structural formula of simvastatin
Other drugs belonging to the class of statins and derived from simvastatin are:
-lovastatin (Lovinacor®, Rextat®);
-pravastatin (Prasterol®, Selectin®, Aplactin®, Pravaselect®, Selectin®, Vasticor®);
-atorvastatin (Torvast®, Totalip®, Xarator®);
-rosuvastatin (Simestat®, Provisacor®).
These drugs differ substantially from each other in duration of action and side effects, however they share the same mechanism of action, namely the inhibition of the enzyme HMG-CoA reductase. HMG-CoA reductase is a hepatic enzyme that represents the limiting stage of the metabolic pathway that leads to the endogenous production of cholesterol.
Simvastatin was discovered and developed by the pharmaceutical company Merk. It was possible to obtain it from the fermentation products of the mushroom Aspergillus terrestris.
Simvastatin is on the World Health Organization (WHO) Essential Medicines List, which indicates the most important medicines needed in a primary health care system.
Indications simvastatin
Simvastatin is indicated for the treatment of primary hypercholesterolaemia or mixed dyslipidaemia, as an adjunct to diet, when the response to diet or other non-pharmacological treatments (such as exercise, weight reduction) is inadequate. Furthermore, simvastatin finds its use for the treatment of homozygous familial hypecholesterolemia in association with a low-fat diet and other lipid-lowering treatments.
Simvastatin can finally be used for cardiovascular prevention, reducing cardiovascular mortality or morbidity in patients with manifest atherosclerotic cardiovascular disease or diabetes mellitus, with normal or increased cholesterol levels, as an adjunct for the correction of other risk factors and other cardioprotective therapies .
Dosage
The dosage range is 5-80 mg / day administered orally as a single tablet dose in the evening.
Contraindications to the use of simvastatin
Simvastatin is contraindicated in case of hypersensitivity to the drug or to the excipients of the tablet, in case of active liver disease or persistent elevations of serum transaminases without obvious cause.
Concomitant intake of simvastatin with the antifungals itraconazole and ketoconazole, with HIV protease inhibitor drugs used for the treatment of AIDS, and antibiotics erythromycin and clarithromycin are contraindicated.
Simvastatin should not be used during pregnancy and lactation.
Side effects
The frequencies of the following adverse effects, reported in clinical trials or post-marketing use of simvastatin, are ranked based on the assessment of their incidence rates across large long-term placebo-controlled clinical trials, including HPS studies and 4S with 20,536 and 4,444 patients controlled respectively.
The frequencies of adverse effects are sorted according to the following criterion: very common (> 1/10), common (> 1/100, <1/10), uncommon (> 1 / 1,000, <1/100), rare ( > 1 / 10,000, <1 / 1,000), very rare (<1 / 10,000).
-Skeletal muscle and connective tissue system: myopathy, rhabdomyolysis, myalgia, muscle cramps (rare);
- Blood and lymphatic system: anemia (rare);
- Nervous system: headache, paraesthesia, dizziness, peripheral neuropathy (rare);
- Gastrointestinal system: constipation, abdominal pain, bloating, dyspepsia, diarrhea, nausea, vomiting, pancreatitis (rare);
- Hepatobiliary system: hepatitis, jaundice (rare); liver failure (very rare);
-Skin and subcutaneous tissue: rash, itching, alopecia (rare);
Evidence of efficacy of simvastatin
The efficacy of simvastatin was demonstrated in Scandinavian Simvastatin Survival Study (Scandinavian Simvastatin Survival Study), a randomized, placebo-controlled clinical trial conducted in a sample of 4,444 people in Scandinavia. During this study project, which lasted 5 years, the intake of 20-40 mg of simvastatin per day produced a reduction of about 35% in LDL cholesterol (bad cholesterol associated with the formation of atherosclerotic deposits on the arterial walls), accompanied by 'increase of about 8% in HDL cholesterol (the good cholesterol).
Simvastatin therapy reduced the overall mortality of 30% in people with pre-existing cardiovascular disorders and high LDL cholesterol levels, and reduced cardiovascular-related mortality of 42%. The drug also reduced the risk of 37% heart attack, 28% stroke, and the need for 37% coronary revascularization procedures.
I study Heart Protection Study (Heart Protection Study) evaluated the effects of simvastatin in people with risk factors, including pre-existing cardiovascular disease, diabetes, or stroke, but with relatively low LDL cholesterol levels. In this study, which lasted 5.4 years, on a sample of 20,536 people, overall mortality was reduced by 13% and cardiovascular-related mortality dropped by 18%. People who took the drug suffered 38% fewer non-fatal heart attacks and 25% fewer strokes.
Bibliographic sources
–Liao JK, Laufs U. Pleiotropic effects of statins. Annu Rev Pharmacol Toxicol. 2005; 45: 89–118.
Articles that may interest you:

